They are found in the serum and tissue fluids of all mammals in the form of a secretion (ANTIBODIES) or bound to the membrane of B lymphocytes (BcR RECEPTOR). They are the mediators of the humoral response. Four types of immunoglobulin have been found in pigs. These are:
IgM, IgG, IgA and IgE. | | HOW ARE IMMUNOGLOBULINS PRODUCED?
Immunoglobulins are produced after stimulation of a B lymphocyte by T dependent or T independent antigens and its subsequent transformation into a plasma cell. The plasma cells do not divide, neither does the isotype change, and they do not express SLA II or immunoglobulins on their surface. They therefore cannot interact with any type of antigen. They are factory cells of immunoglobulins. |
|
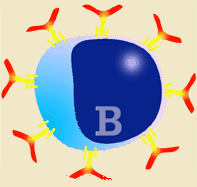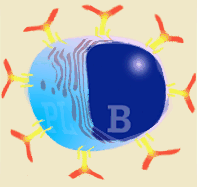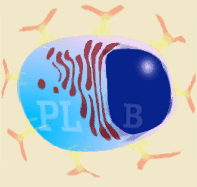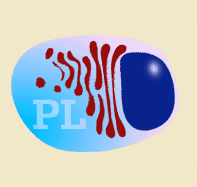
Diagram of B lymphocyte and its transformation into a plasma cell
|
Given the enormous structural complexity of most antigens, when a humoral response is produced against them a high number of different antibodies is induced targeting the various epitopes that make up the antigen which induces the immune response. The humoral immune response is therefore polyclonal, as there are thousands of stimulated clones secreting different antibodies.
| The immune response mediated by antibodies is called a humoral response. |
| The humoral response is polyclonal and can be a primary or secondary response |
The immunoglobulins produced by each plasma cell clone are specific against the epitope which induces an immune response. There are as many clones producing antibodies as there are epitopes that have induced an immune response. In a response induced by an antigen (made up of several epitopes) there are various plasma cell clones secreting antibodies specific to each epitope.
|
|
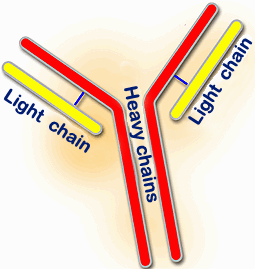
The basic structure of immunoglobulins made of two heavy chains (red) and two light chains (yellow) |
WHAT IS THE STRUCTURE OF IMMUNOGLOBULINS?
The structure of pig immunoglobulins, as in other species, is based on glycoproteins which mainly consist of four polypeptide chains. Two of these are named HEAVY or H chains and have a molecular weight of 55 to 77 kilodalton (kDa). The remaining two are named LIGHT or L chains and have a molecular weight of 23 to 26 kDa. Each member of a pair of chains is identical in structure to the other. The two heavy chains bind to each other covalently by means of a disulphide bond and heavy and light chains are also bound by a disulphide bond. Each chain has a constant and a variable region. |
|
If an immunoglobulin molecule is treated by proteases, such as pepsin or papain, it splits into two fragments known as Fab, an acronym for "antigen binding Fragment" and Fc (Crystallizable Fragment). The specificity of immunoglobulin resides in the first fragment (Fab) and also in its capacity to react with the antigen, whilst the second fragment (Fc) carries out the effector functions of the immunoglobulins (complement fixing, cell receptors, etc).
Both heavy and light chains consist of reserved protein structures known as "immunoglobulin domains". These domains are made up of approximately 110 amino acids. The light chains consist of two domains, one variable (VL) and the other constant (CL). The heavy chains have one variable part (VH) and three (IgG and IgA) or four (IgM and IgE) constant parts (CH1, CH2, CH3 and CH4). The domains are identical between the two light chains and between the two heavy chains.
|
|
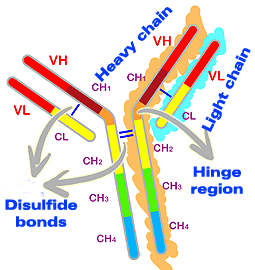
Structure of immunoglobulin domains. The light chains consist of two domains, one variable (VL) and the other constant (CL). The heavy chains present one variable (VH) and three or four constant (CH) domains.
|
In the H chains there is an additional region named the hinge region which does not form part of the domains. The hinge region is located between the CH1 and CH2 domains and permits the mobility of immunoglobulins. Analysis of amino acids from the hinge region shows that it contains a large amount of the amino acid proline. This gives it flexibility, but also susceptibility to attack by proteases, leading to Fab and Fc fragmentation.
The variable domains (VL and VH) have the task of antigen binding and are therefore responsible for the specificity of immunoglobulin. The constant domains permit the differentiation of five heavy chain isotypes (m,g,e,a,d) which make up the immunoglobulins (IgM, IgG, IgE, IgA and IgD) and two types of light chains: kappa (K) and lambda (l). They are responsible for the effector functions of immunoglobulins (complement fixing, cell receptors, etc). |
|
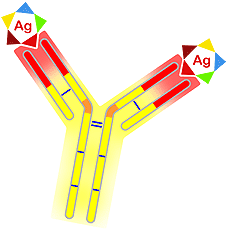
Location of antigen binding sites |
The variability observed in the variable areas of both chains (L and H) are located in three segments with around 10 amino acids. These are named hypervariable regions, and also known as CDR1, CDR2 and CDR3 (Complementary Determining Regions). These segments make up the aforementioned antigen binding site. Each immunoglobulin molecule has two antigen binding sites.

|
|

Diagram of the citotoxicity reaction mediated by antibodies. Specificity is provided by the antibody and the cell's cytotoxic action |
Carbohydrates also play an important role in the structure of immunoglobulins, above all in the constant region of the heavy chains, and in particular in the CH2 area and in the hinge region, even though carbohydrates only represent around 15% in the variable regions of the heavy chains. The role of sugars is not at all clear, but seems to be linked to catabolism and also affects some functions. It has been proved that deglycosylated IgG lose or reduce their capacity to bind to the cell receptors and induce ADCC, as well as to activate the complement. |
|

